A) CH3CH2CH2OH
B) CH3C≡CCH2CH3
C) CH3CH2CH(NH2) CH3
D) (CH3) 3N
E) CH3CO2CH2CH3
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which color is observed when a sample absorbs light at 650 nm?
A) yellow
B) orange
C) red
D) violet
E) greenish-blue
G) A) and C)
Correct Answer

verified
Correct Answer
verified
Essay
Is the λmax of phenol in aqueous solution affected by the pH of the solution? If so, explain how and why λmax changes.
Correct Answer

verified
As the pH of the solution increases, an ...View Answer
Show Answer
Correct Answer
verified
View Answer
Multiple Choice
Which compound would be expected to show intense IR absorption at 1715 cm-1?
A) CH3CH2CO2H
B) 1-hexene
C) 2-methylhexane
D) CH3CH2CH2NH2
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Short Answer
2-Methylhexane shows an intense peak in the mass spectrum at m/z = 43. Propose a likely structure for this fragment.
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Identify the compound that has a formula of C3H6O with a stretching vibration at 1050 cm-1 and an absorption band at 3400 cm-1, but no doublet at 1380 cm-1.
A) 1-propanol
B) 2-propanol
C) ethyl methyl ether
D) propen-1-ol
E) propen-2-ol
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Ethyl acetate has the structure CH3CO2CH2CH3. Which set of m/z peaks is most consistent with this structure?
A) 88, 73, 43
B) 88, 76, 45
C) 88, 73, 45
D) 88, 76, 43
E) 88, 72, 70, 44
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Predict the molecular formula of the compound represented below based on the MS data given. 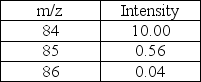
A) C6H12
B) C5H24
C) C4H6O2
D) C3H8O2
E) C5H8O
G) B) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
How many centimeters (cm) are there in one micrometer (μm) ?
A) 10-6
B) 10-3
C) 10-5
D) 10-8
E) 10-4
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following m/z values is the molecular ion for 2-butanone? 
A) 15
B) 29
C) 43
D) 57
E) 72
G) B) and C)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following is not a common bending vibration of a CH2 group?
A) flip
B) rock
C) scissor
D) twist
E) wag
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following solvents is best used in infrared spectroscopy? 
A) I
B) II
C) III
D) IV
E) V
G) A) and B)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
The mass spectra of alcohols often fail to exhibit detectable M peaks but instead show relatively large ________ peaks.
A) M+1
B) M+2
C) M-16
D) M-17
E) M-18
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the mass spectrum of 2-chloropropane, what is the m/z of the molecular ion peak of greatest intensity?
A) 63
B) 76
C) 78
D) 80
E) 82
G) None of the above
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which compound would be expected to show intense IR absorption at 2710 and 1705 cm-1?
A) CH3COCH2CH3
B) PhCOCH3
C) PhCHO
D) CH2 ![]() CHCOCH3
CHCOCH3
F) B) and D)
Correct Answer

verified
Correct Answer
verified
Essay
Provide the structure of the two ions which result when the molecular ion of 2-methoxypentane undergoes fragmentation by 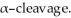
Correct Answer

verified
+ +
CH3CH2CH2CH  _TB1830...
_TB1830...View Answer
Show Answer
Correct Answer
verified
CH3CH2CH2CH
View Answer
Multiple Choice
Which molecule below has two significant bands in the IR at 2720 cm-1 and 2820 cm-1 (medium) ?
A) CH3COCH2CH
B) CH3CH2CO2CH3
C) CH2=CHCH2CH3
D) CH3CH2CH2CHO
E) CH3CH2CH2OH
G) C) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
In the mass spectrum of bromoform, CHBr3, molecular ion peaks appear at m/z 250, 251, 252, and 253? What is the ratio of the intensities of these peaks?
A) 1:1:1:1
B) 1:2:2:1
C) 1:3:3:1
D) 2:3:3:2
E) 3:2:2:3
G) A) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which of the following statements is not true about electromagnetic radiation?
A) The velocity of light is directly proportional to the energy.
B) All molecules absorb electromagnetic radiation at some frequency.
C) Frequency is inversely proportional to wavelength.
D) Energy is directly proportional to frequency.
E) Energy is inversely proportional to wavelength.
G) D) and E)
Correct Answer

verified
Correct Answer
verified
Multiple Choice
Which molecule below has a significant band in the IR at 3300 cm-1 (medium and sharp) ?
A) 1-pentene
B) cyclohexane
C) 2-heptyne
D) 1-pentyne
E) diethyl ether
G) C) and D)
Correct Answer

verified
Correct Answer
verified
Showing 81 - 100 of 127
Related Exams